
RedHill Biopharma Ltd., a specialty biopharmaceutical company, developed RHB-204, a treatment for MAC, and needed help streamlining the process of getting into Phase 3 clinical trials. RHB-204 combines three well-known antibiotics: clarithromycin (CLR), clofazimine (CFZ), and rifabutin (RFB). The effects of the three individual antibiotics as well as two-drug combinations have been described in several non-clinical models of NTM systemic and lung infection. However, there was no model available for the combination of the three antibiotics. tags so 3
- In Vitro Hollow Fiber Study. In collaboration with Dr. Tawanda Gumbo from Praedicare Inc, Certara scientists supported the design and interpretation of an in vitro lung infection hollow fiber study (HFS) to evaluate the pharmacokinetics and behavior of a drug in a controlled and reproducible manner. This assay can be used to help optimize dosing regimens, reducing the number of animal and human trials required to evaluate a drug’s safety and efficacy.4
The HFS-MAC was used to assess the contribution of each individual drug component of RHB-204 (CFZ, CLR, and RFB) when administered individually and as part of a two or three-drug combination. The study was also instrumental in determining anti-mycobacterial effects and understanding how the drugs react when used as part of a combination regimen
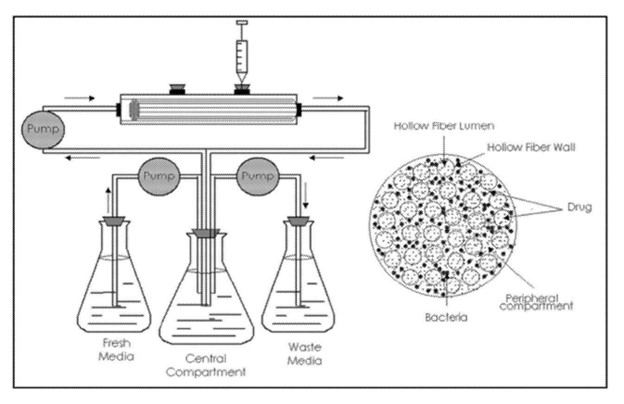
Figure 1. In vitro hollow fiber study.
- Inderlied CB, Kemper CA, Bermudez LE. The Mycobacterium avium complex. Clin Microbiol Rev 1993;6(3):266-310. doi:10.1128/CMR.6.3.266. PMID: 8358707; PMCID: PMC358286. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC358286/.
- Akram SM, Attia FN. Mycobacterium Avium Intracellulare. [Updated 2022 Apr 2]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK431110/.
- Fathi et al. Triple antibiotic fixed-dose combination products, dosing regimen, methods, and kits for treating pulmonary non-tuberculous mycobacterial infections. United States Patent Application Publication. Pub. No.: US 2021/0401865 A1. Pub. Date: 2021 December 30. [Internet]. [cited 2023 Feb 09].
- Nenortas E, Bakshi RP, Shapiro TA. Hollow-fiber methodology for pharmacokinetic/pharmacodynamic studies of antimalarial compounds. Curr Protoc Chem Biol. 2016 Mar 16;8(1):29-58. doi:10.1002/9780470559277.ch150194. PMID: 26995353; PMCID: PMC4811375. [Internet]. [cited 2023 Feb 09]. Available from: https://doi.org/10.1002/9780470559277.ch150194.
- Lebeaux D, Chauhan A, Rendueles O, Beloin C. From in vitro to in vivo models of bacterial biofilm-related infections. Pathogens. 2013 May 13;2(2):288-356. doi:10.3390/pathogens2020288. PMID: 25437038; PMCID: PMC4235718. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4235718/
Contact us





