Kimiko McGirr
Scott Gruver
Diana Marcantonio
April 4, 2025
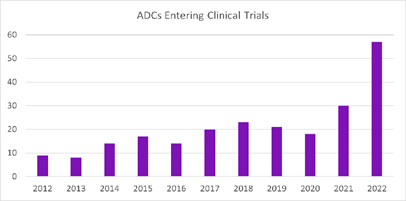
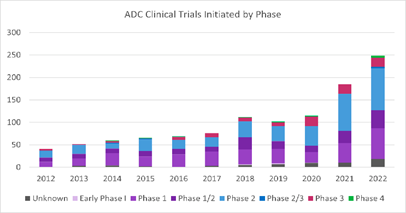

QSP model pack for antibody-drug conjugates
Pre-built library of validated QSP models for ADCs with and without avidity
Learn more about our QSP Model Pack for Antibody-Drug Conjugates (ADC)
Includes 42 models for monospecific and bispecific ADCs
Features avidity effects and multi-compartment pharmacokinetics
Supports in vitro and in vivo simulations

Download the ADC model pack
Discover how Certara’s Antibody-Drug Conjugate (ADC) Model Pack in AssessTM can transform your ADC research.






